Protein Modeling of S-Naproxen
from Human Serum Albumin
Hamna Riaz
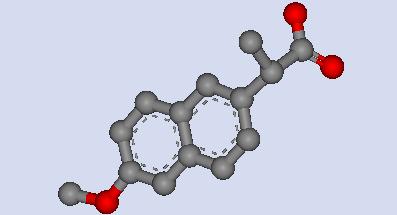 |
This is 3D molecular model of s-naproxen, a heterocompound complexed within human serum albumin. The molecular formula is C14H13O3 |
 |
This is the Lewis structure for s-naproxen. The three oxygens within the compound each have non-bonding electrons. The acidic property of this compound can be noted by the negative charge on one of the oxygen substituents. |
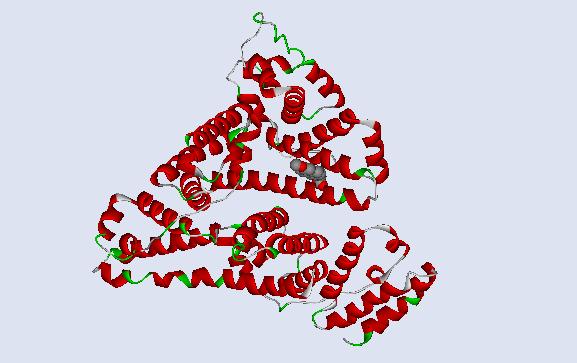 |
This is a 3D ribbon representation of the protein human serum albumin. S-naproxen is in the CPK form so that its location relative to the entire structure can be clearly observed. |
 |
This is a depiction of the two superimposed structures of s-naproxen before and after minimization. The most prominent change is between the conformation of the carboxylic acid substituents of both structures. |
Part II
Now the the structure for s-naproxen and its energy properties have been explored, the interactions that contribute to its structure will be analyzed. To view the full analysis with diagrams please see the html format of the word document.
Click here to see my word document on the analysis of the heterocompound s-naproxen.
Click here to see an html version of the word document above
Click here to see the minimized conformation of decanoic acid, viewable in Chem 3D
References
The structure for s-naproxen was obtained from the following web sites:
References:
[1]
A. Yamaguchi, K. Iida, N. Matsui, S. Tomoda, K. Yura, M. Go: Het-PDB Navi. : A database for protein-small molecule interactions. J. Biochem (Tokyo), 135, pp.79-84 (2004)
[2]
Lejon,
S., Cramer,
J.F., Nordberg, P.A. ![]() (2008)
Structural Basis for the Binding of
Naproxen to Human Serum Albumin in the Presence of Fatty Acids and the
Ga
Module.
(2008)
Structural Basis for the Binding of
Naproxen to Human Serum Albumin in the Presence of Fatty Acids and the
Ga
Module. ![]() Acta
Crystallogr.,Sect.F
Acta
Crystallogr.,Sect.F ![]() 64: 64
64: 64 ![]()
DOI 10.2210/pdb2vdb/pdb
[3]
S.Lejon et al. (2008). Structural basis for the binding of naproxen to human serum albumin in the presence of fatty acids and the GA module.. Acta Crystallogr Sect F Struct Biol Cryst Commun, 64, 64-69. [PubMed id: 18259051] [DOI: 10.1107/S174430910706770X] (abstract only, see [8])
[4]
Chem 3D Help, “Compute Properties.”
[5]
"Naproxen." AHFS Consumer Medication Information. 2009. American Society of Health-System Pharmacists. 4 May 2009 <http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=meds&log$=drug_bottom_one∂=a681029>.
[6]
Block Drug Co., Inc., Jersey City, NJ 07302; Hazleton Laboratories America, Inc., Vienna, VA 22180; Department of Pharmaceutical Sciences, College of Pharmacy and Allied Health Professions, St. John's University, Jamaica, NY 11439 [DOI: 10.1002/jps.2600770314]
[7]
MedInnovation GmbH: function of Human Serum Albumin. Retrieved May 4, 2009, from http://www.medinnovation.de/background/hsa.htm
[8]
For
Full Text
Article:
S.Lejon et al. (2008). Structural
basis for the binding of naproxen to human serum albumin in the
presence of
fatty acids and the GA module.. Acta Crystallogr Sect F
Struct Biol Cryst
Commun, 64, 64-69. [DOI:10.1016/j.physletb.2003.10.071]
[9]
PDBSum: HSA complexed with S-naproxen structure. Retrieved May 5, 2009, from http://www.ebi.ac.uk/thornton-srv/databases/cgi-bin/pdbsum/GetPage.pl?pdbcode=2vdb&template=main.html
Back to the Chem 350 Projects Page